New $1.25 million grant enables investigation into vital role of sex hormones in tissue repair
LAWRENCE — Our ability to heal and regenerate new tissue after an injury has much to do with biological sex. For instance, following menopause, the loss of sex-hormone signaling in women can lead to degenerative diseases such as osteoporosis and osteoarthritis, where bone and cartilage degrade and become more delicate and brittle because of tissue loss.
Now, a researcher at the University of Kansas has earned a five-year, $1.25 million Maximizing Investigators’ Research Award from the National Institutes of Health to study how estrogen interacts with the body at the level of the cell microenvironment. The results could point the direction for the study of sex differences in human mesenchymal stromal cells (MSCs), specialized cells that can support a host of other cells by promoting tissue repair and regeneration after injury and represent a key to developing new tailored therapies.
“I’m trying to understand why male and female tissue might regenerate differently and how this leads to the onset of diseases where the tissue does not regenerate well, such as osteoporosis and osteoarthritis,” said Jenny Robinson, assistant professor of chemical & petroleum engineering at KU, who will lead work under the NIH MIRA award. “We’re going to approach this question at the level or microenvironment that cells live in, essentially, and see how that might dictate regeneration. We’re looking at estradiol, the most predominant estrogen, and coupling that knowledge with control of the cells’ 3D environment to see, ultimately, what do we need to promote repair, and that is likely different in male and female tissues.”
 As a teen athlete, Robinson was sidelined from pursuing a serious soccer career because of a knee injury, but that also triggered her interest in the science involved in the body’s recovery, a curiosity that led to her expertise in biomaterials, tissue engineering and biomolecular engineering. “My lab is really focused on connective tissue,” she said. “We do a lot with the knee meniscus. It’s really what we’ve been doing for the past three or four years.”
As a teen athlete, Robinson was sidelined from pursuing a serious soccer career because of a knee injury, but that also triggered her interest in the science involved in the body’s recovery, a curiosity that led to her expertise in biomaterials, tissue engineering and biomolecular engineering. “My lab is really focused on connective tissue,” she said. “We do a lot with the knee meniscus. It’s really what we’ve been doing for the past three or four years.”
The KU researcher’s new work aims to develop biomaterial tools to parse out sex differences in tissue repair and how bodies maintain homeostasis, or a necessary equilibrium to promote health and function after changes such as injury. The work will follow three main paths:
First, Robinson hopes to understand better how male and female MSCs respond differently to spatial and mechanical properties of the cell microenvironment using engineered biomaterials in her lab.
“There’s some clinical data to show there might be differences in structural tissue components in men and women,” she said. “We know cells are very responsive to their structural environment, and in these engineered systems we can control a lot of factors. My lab builds polymer-based 3D structures that mimic tissue. We use a technique to generate fibers on the scale of collagen — so nanometer to small micron scale — and know that across the board cells respond completely differently if they’re on a nanometer versus a micrometer fiber, or if those fibers are just randomly oriented or aligned like in muscle or a lot of connective tissue such as the meniscus. But what if they’re from male or female patients — are those cells responding differently to the structural cues that we can engineer and provide for them in the lab?”
Additionally, Robinson aims to discover how estrogen affects downstream transcription, or how cells copy segments of DNA into RNA to make essential proteins, and cells’ subsequent behavior. To do so, her lab has developed customized microchips that expose cells to different concentrations of estrogen.
“We’ve adapted previous designs and fabricated at the KU Nanofabrication Facility. They’re easy to make and fairly cheap,” Robinson said. “Researchers are showing estrogen has effects through a wide range of estrogen concentrations and so depending on the concentration, you could say, ‘Well, estrogen's doing this, or it’s doing the opposite.’ If you say, ‘I’m going to look at this range of concentration, and I’m going to manually do it,’ it’s going to take a lot of time. We’re looking at nine orders of magnitude change in concentration. However, we can address this large range of concentration in these devices by seeding cells — you just need a small amount — and then the devices are able to quickly apply different concentrations through a lot of channels using diffusion. You get a range of concentrations just by mixing from zero to a high concentration through these channels and create a gradient of concentration. The idea is you dose these cells for 24, 48 or 72 hours, then you can take the cells out and start to look at how the cells are responding based on these concentrations. These microchips will save on the time and cost needed to complete these studies.”
Finally, Robinson and her lab hope to use their results to engineer biomaterial systems that effectively regulate the presentation of estrogen to cells, an advance that could lead to more effective treatments for a host of injuries and chronic conditions that damage bones and connective tissue.
“The way we make biomaterials, we can control the release of estrogen. Much like slow-release pills, we can take the idea of microspheres and drug formulation but take it to the 3D scaffold material,” she said. “We actually can make a biomaterial kind of closer to product where you say, ‘This is the structure I need for the cells to respond, and it releases either this hormone or hormone modulator at this rate that we know is really important for repair.’”
While the research initially centers on estrogen and potential biomaterials geared toward women, the KU researcher said her investigation could result in therapies that benefit men as well.
“If someone is going to be put on an aromatase inhibitor — for instance, men are on these inhibitors to prevent testosterone conversion to estrogen, this will have implications for both biological sexes,” she said.
Robinson said that work under the grant also will help train the next generation of scientists in the field of engineering biomaterials.
“I have 14 people in the group right now, including undergrads, grad students and a postdoc, and they’re doing different projects all encompassed in this work,” she said. “For me, the training and the learning process for the students is my favorite thing. I love the ability to give a big-picture idea and have them run with it. The ability to mentor these trainees is one of the main reasons why I love academic research.”
Yet, Robinson said the end goal is to carry out research that will improve people’s health.
“So many people have to deal with musculoskeletal injuries,” she said. “It affects people’s quality of life. It’s one of the biggest issues that causes disability and unemployment and lack of being able to work in general. Orthopedic injury and degeneration are likely to plague many people. Everyone knows someone that has injured some type of connective tissue.”
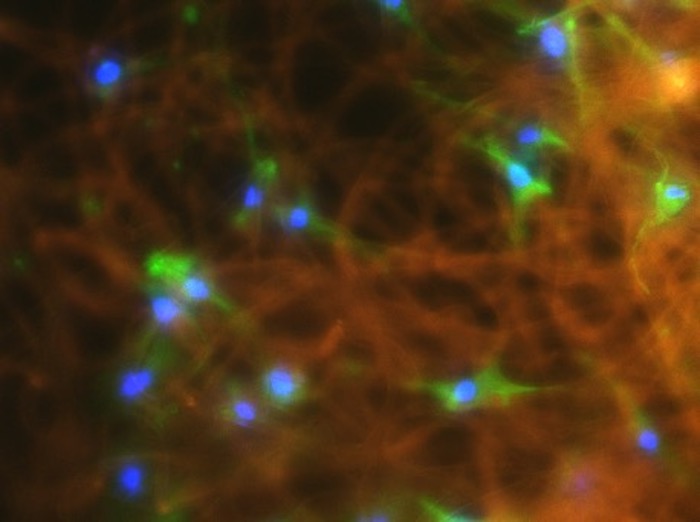
Top photo: Human mesenchymal stromal cells on fibers. Credit: Jenny Robinson.
Right photo: Well plates for mesh in Robinson Lab. Credit: Jenny Robinson.